Oncology
Advancing Oncology Commercialization
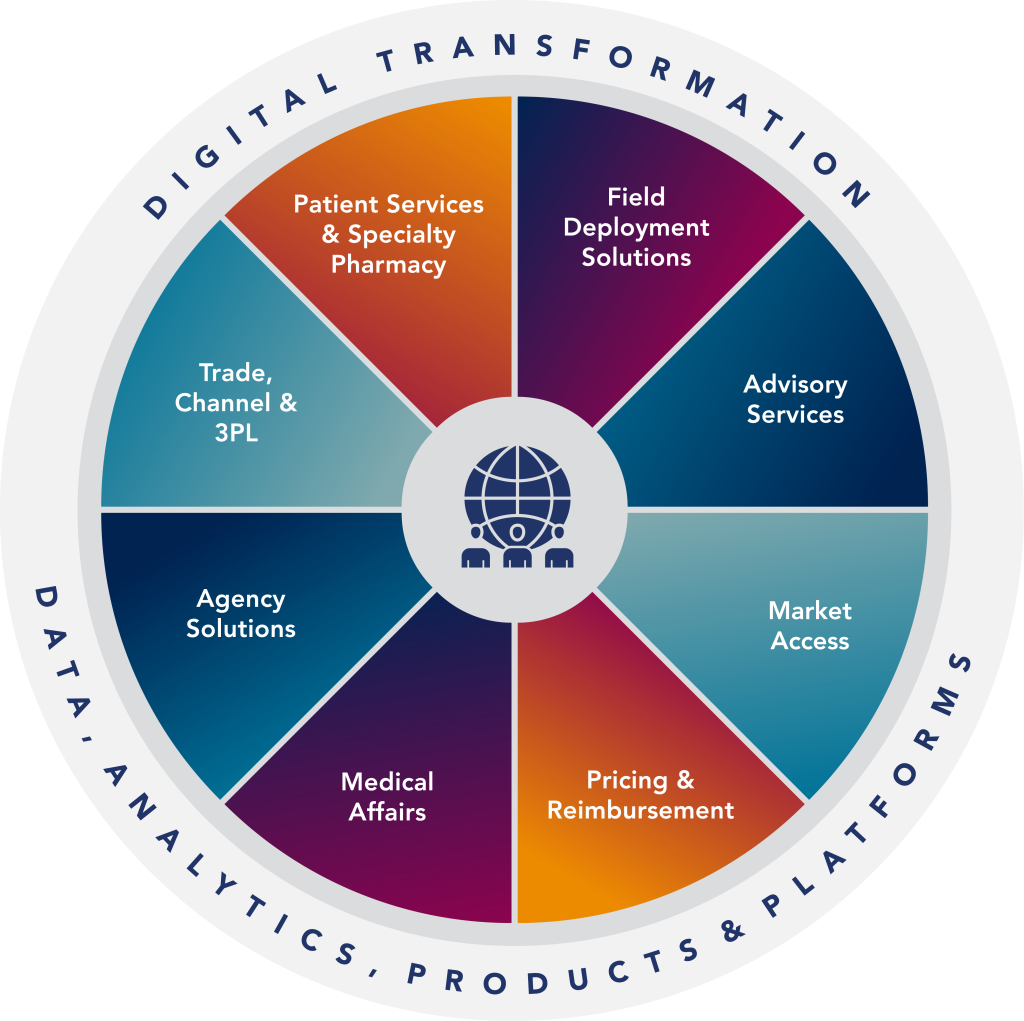
Delivering Impact Through Oncology Expertise and Innovation
Where Expertise Meets Innovation
Expertise
Oncology experts driving commercialization—from strategy to patient services.
Innovation
Reimagining oncology commercialization through AI, data, and emerging technologies.
Meet with Our Change Makers

Anne Marie Robertson
EVP, EVERSANA ONCOLOGY®

Suzanne Greenwood, RN, BSN
Chief Commercial Officer

Tim Lewis
Strategic Development, Agency Services

Barry Vucsko
SVP, Strategic Development, Agency Services

Nick Rambke
SVP, Client Services, Agency Services

Gurdip Daffu, PhD
SVP, Medical, Agency Services

Vanitha Sankaran, PhD
SVP, Medical, Agency Services

Susan Giacalone, RN
Vice President, Medical Deployment

Jordan Schell
Associate Partner, Advisory Services

Jennifer Bartosh
Senior Director of Training, Field Solutions

Pierantonio Russo, MD, FCPP, FAAP, STS
Chief Medical Officer
Engage with Oncology Innovators
Partner with experts transforming commercialization through deep therapeutic insight.
Integrated Expertise. Data-Driven Innovation. Impactful Oncology Solutions
EVERSANA ONCOLOGY delivers a unified, expert-led approach to commercialization, combining deep therapeutic knowledge with advanced technology platforms to solve challenges others haven’t even asked. Our data-driven strategies and measurable outcomes empower partners to navigate the complexities of oncology with confidence and clarity.


Accelerating Oncology Commercialization with Proven Expertise
From early development to brand revitalization, EVERSANA ONCOLOGY delivers agile, customized solutions backed by a track record of success across 130+ products and 45+ companies.

Reimagine Oncology Commercialization
Discover how our expert-driven, AI-powered solutions are transforming the way life sciences partners navigate the complexities of oncology.
Oncology Expertise & Insights
Case Studies: Real-World Impact
Leveraging our data and analytics platform, our team works together to produce the best outcomes for our partners and their patients.
It’s Time to #OvaryAct
Awareness about ovarian cancer was lacking. We changed that in a big way.
Accelerate Your Oncology Impact
Contact our oncology experts to get started.