Insights
Integrated Solutions
Rooted in the Patient Experience
Across the Product Life Cycle
Related Articles

Successfully Navigating Self-Commercialisation in Europe
Introduction As discussed in our previous paper, Successful Launches in Europe: Complex, But Not Complicated?,, Europe is one of the world’s largest and most important regions of focus for pharmaceutical and biotechnology manufacturers looking to treat patients and maximise the value of their products. However, the challenges of entering this market, particularly in terms of […]

Successful Launches in Europe: Complex, but Not Complicated?
Europe represents a major pharmaceutical market – it accounts for 22% of the global market, second only to the U.S. in terms of market size and has a large population of over 500 million. Understandably, pharmaceutical and biotechnology companies looking to maximise their revenue consider Europe to be a key focus[1]. Indeed, looking at first-time […]

The Future of Pharma – A Provocative Perspective
In this episode of AI For Pharma Growth, Dr. Andree Bates is joined by Mike Ryan, Vice President, Europe, EVERSANA. They discuss the future of pharma and the impact of recent advancements in healthcare. The industry is facing a unique moment, with technology opening up new possibilities for medical interventions and a greater focus on […]

How Specialty Pharmacies Can Elevate Your Channel and Distribution Strategy for HCP-administered Buy & Bill Products
A successful channel and distribution strategy for Buy & Bill (B&B) products should be centered around the partners you sell to and through. That means considering any third-party logistics (3PL) providers, specialty distributors (SD) and specialty pharmacies (SP) who will touch your product between the manufacturing/packaging site and patient administration. Taking a holistic view of […]

New Medicines, New Access, New Frontiers
How psychedelics, cell therapies and other innovations are driving regulatory reform and new patient access Science is improving exponentially. What’s now possible, through new medicines, and entirely new categories of medicine, could only be dreamt of just a decade ago. Doctors and patients are naturally excited about the oncoming pipeline. But while science improves exponentially, […]

Impact of Patient Reported Outcome Measures on HTA Decisions for Rare Diseases
As rare diseases become an increasing area of global focus, pharmaceutical manufacturers are taking a closer look at how patient-reported outcomes (PROs) may be used to improve uptake in HTA evaluations. Although “rare” suggests not many people are affected with a condition, in the EU between 6,000 and 8,000 different rare diseases affect an estimated […]

What You Need to Know as the European Union Embarks on Joint Health Technology Assessment (HTA)
Facing the “Fourth Hurdle” Member states of the European Union benefit from a centralised marketing authorisation process for medicinal products. Also, since its implementation in 1993, the European Medicines Agency (EMA) has assured pharmaceutical companies the right to commercialise products that underwent a centralised marketing authorisation in the second largest (and most diverse) single pharma […]

How Do We Pay for a Cure? How to Put a Price on Life-changing Treatments
The global cell and gene therapy market is expected to reach $13.8 billion by 2026, expanding at a compound annual growth rate of 12.4%. — Global Cell & Gene Therapy Manufacturing Services Market by Type – Forecast to 2026 Report With over 2,000 ongoing clinical trials in regenerative medicine, patients across the world will soon […]
How to Ensure Speed and Efficiency When Launching in Europe
Today is an extraordinary time for medical innovation. The pharmaceutical industry is yielding protection against diseases, focusing more on rare and complex disease treatments, and the world is getting better health outcomes. Global life expectancy is more than 70% higher today than it was in 1960 as diseases that were once considered terminal are now […]

Considering Commercial Success During Clinical Development: Maintaining a Global Perspective
Although commercial success is the ultimate goal of pharmaceutical research and development (R&D), many new treatments do not meet expectations and fail to achieve broad global reimbursement after initial regulatory approval., This causes therapies to underperform across international markets and, more importantly, limits access for patients in need. For example, only 56% of all new drugs approved by the European Medicines Agency (EMA) […]

EPP Keynote Address: Global Pricing and Market Access Hurdles for Innovation
Global Pricing and Market Access Hurdles for Innovation In a recent keynote address at the 2021 EPP Life Sciences Pricing Conference, Alan Crowther, General Manager, EVERSANA Pricing and Market Access, discusses 4 hurdles that are serving as obstacles to innovation in global pricing and market access including: Increased pricing pressures Changing launch dynamics that impact […]

Pharma Europe 2021 : Panel
A Data and Digital Enabled GTM Strategy for Long-term Business Impact Rohit Sood, Executive Vice President, COMPLETE Commercialization, joined a panel of industry experts to discuss why an aligned and cross-functional go-to-market strategy has never been more important and how to balance strategic priorities in this Pharma 2021 conversation. Watch the recording: Meet with our experts.

Bio-Europe “Collaboration Close-Up”: Exploring the EVERSANA – Shorla Pharma Partnership
Launching in today’s unpredictable market is a journey with new challenges around every corner. While the oncology pipeline is rapidly growing, stringent competition and an evolving provider environment are putting intense pressure on manufacturers to build effective commercialization infrastructures and launch products at unprecedented speeds, which comes at a steep price. In this session, Shorla […]
WEBINAR: Yes EU can! How novel treatments can avoid complexity and commercialize with impact across the European continent
Europe has a population of more than 450 million people, yet most manufacturers consider the U.S. the primary market for launch. In this webinar, EVERSANA’s Mike Ryan, Executive Vice President, Europe, discusses with other industry leaders how the EU’s landscape is changing in order for European states to increasingly become a priority market for commercialization. This […]

EPP Studio Live Webinar — Impact of COVID-19 Pandemic on German Healthcare System
Leading up to the recent German elections, the topic of healthcare took center stage for the aging German population. With nearly 90% of the population participating in Statutory Health Insurance (SHI), increased financial pressures have widened the gap between SHI coverage and anticipated expenditures. During this EPP Studio Live Webinar, Douglas Foerster, Senior Vice President […]

EPP Studio Live Webinar — EU Joint Procurement of COVID-19 Vaccines: A Model for Future Market Access in Europe?
The COVID-19 pandemic has changed the way we live and the way we respond to global healthcare crises. During this EPP Studio Live webinar, Douglas Foerster, Senior Vice President of Pricing & Market Access for EVERSANA, shares a brief historical summary of how the European Union negotiated the acquisition of COVID-19 vaccines through an unprecedented, […]

WEBINAR: The New Path to European Pharmaceutical Commercialisation
Global healthcare and life sciences innovation continues to surge at an unprecedented rate. External disrupters such as Brexit, GDPR, new regulatory guidance, such as the Pharmaceutical Strategy for Europe, and the COVID-19 pandemic, are driving pharma and healthcare leaders to reevaluate long-standing strategies. If pharma companies want to remain globally relevant and competitive, they must reassess commercialisation strategies – particularly related to clinical […]

EPP Studio Live Webinar — Looking Ahead: Landscape of the Key Price and Market Access Trends in Europe
The global healthcare landscape continues to change at a rapid pace. As a result, new pricing and access trends are emerging across the EU and beyond, driving significant changes in the life sciences industry. During this EPP Studio Live webinar, “Landscape of Key Price and Market Access Trends in Europe,” Alan Crowther, General Manager, Global Products for EVERSANA, shares his insights on […]

Simplifying EU Distribution to Maximize Cost Efficiency and Speed to Market for Patients and Manufacturers
COVID-19 ignited a spark of innovation in the healthcare industry, forcing global markets to reconsider drug development and commercialization processes. The European Union (EU), specifically, is taking carefully planned steps into a new phase of pharma with recent changes, including the Pharmaceutical Strategy for Europe. But one element of the European pharma industry that remains […]

INFOGRAPHIC: What You Need To Know About The Tender Landscape In Europe
In today’s market, manufacturers need real-time pricing insights to remain competitive and develop strategies supported by the right data. PriceRight® by EVERSANA is helping manufacturers manage enterprise and government pricing changes for markets around the world, which is critical as manufacturers launch globally. More specifically, manufacturers launching in Europe must be up to date on regulatory changes, including those related to Brexit […]

The Secret’s Out: The Impact of Net Price Transparency
Pricing system pressures are increasing for the top four markets in Europe. Price transparency, changes to public health plans and curative therapies are set to disrupt traditional drug pricing. To navigate this ever-shifting landscape, companies must carefully consider product adoption and launch sequencing, and understand the potential impact that net price transparency might have. In […]

A New Pharmaceutical Strategy for Europe
In late November, the European Commission announced the adoption of a new healthcare plan – the Pharmaceutical Strategy for Europe. This plan aims to strengthen the European Health Union while ensuring affordable patient access across the entire European Union and supporting sustainable innovation for pharmaceutical industries. What’s Changing, and Why? Until now, patient access and […]

Do Germany’s New Hemophilia Regulations Have Wider-Reaching Implications?
In July 2019, the Drug Safety and Supply Law (GSAV) was passed by Germany’s Higher Chamber of Parliament, bringing about substantial changes to Germany’s health policy. Besides introducing new strategies for biosimilar uptake and cell and gene therapy monitoring, GSAV provides for more stringent control of the distribution of hemophilia products. Download the FULL Pricentric […]

Italy Tackles HTA, Pricing and Reimbursement Reforms
In July, new legislation set to overhaul the way pharmaceuticals are priced in Italy was finally implemented, following an announcement that dates all the way back to August 2019. The provision, which was signed by then Ministers Giulia Grillo (Health) and Giovanni Tria (Economy), has rolled out new criteria and methods by which the Italian […]

eBook – 2020 Stories That Shaped Commercialization
While many industries hit the pause button on their operations this year, the pharmaceutical industry never stopped, propelling new innovations to better serve patients, providers and stakeholders. At EVERSANA, we worked closely with manufacturers to tackle uncertain, complex market dynamics and solve pricing, access, reimbursement, adherence, and distribution challenges, and more. As we look forward […]

NEXT GENERATION COMMERCIALIZATION: A CONVERSATION WITH THE INNOVATORS
Sell, out-license or launch internally: these are the three traditional options to commercialize a pharmaceutical product. Selling and out-licensing are common but cause innovators to lose ownership in an investment that takes years to develop. Launching internally requires an average investment of $125MM+ before the product even hits the shelves. Until now, there was no […]

How to Get Your Digital Therapeutic Approved & Reimbursed in Europe and the U.S.
Patients, providers, and payers expect healthcare to be more accessible, intuitive, and adaptable to their needs. Barriers that have held back innovation in Digital Medicine and Telemedicine have been blasted apart with the recent relaxing of CMS and HHS requirements, continuing advances within the FDA’s Digital Health program, and additional reimbursement opportunities emerging globally. Get […]

The Importance of Digital Strategy in Pharma in the APAC Region
The Asia Pacific region continues to show great progress in digital health as pharmaceutical companies look at digital technologies as a tool to drive access and to start developing their digital strategies. To understand the strides and recent evolution of digital in pharma, our APAC team conducted a survey with top pharmaceutical companies in the […]

Improving Patient Outcomes Through Digital Health
Digital applications can work cohesively with therapeutics in the treatment of a broad range of diseases, from behavioral health to chronic conditions. In an interview with Debraj Dasgupta, Head of Strategy & Go-to-Market Planning at Nippon Boehringer Ingelheim Co., Ltd., Ed Cox, Executive Vice President, Strategic Alliances & Global Head of Digital Medicine at EVERSANA, […]

The Digitalization of Healthcare in Asia Pacific
The digitalization of healthcare in Asia Pacific is being utilized to enhance the patient-centered approach and expanding access to health. In a candid conversation with Abhishek Shah, CEO at Wellthy Therapeutics, Ed Cox, Executive Vice President, Strategic Alliances & Global Head of Digital Medicine at EVERSANA, uncovered the current healthcare landscape and how digital strategy […]

The Regulatory Landscape in Digital Health
Despite the advances in digital health and digital therapeutics in recent years, there remains an ambiguity in the digital space and uncertainty surrounding regulation and reimbursement of digital technologies applied to pharma, biopharma and medical device companies. In an insightful conversation that was part of EVERSANA’s Digital Symposium, Ed Cox, Executive Vice President, Strategic Alliances […]

Access to Technology and the Rise of Digital Health in Asia Pacific
The access to technology, development of telehealth and evolution of digital therapeutics are thriving in Asia Pacific; and pharmaceutical companies are levering the momentum to reach to a broader segment of the patient population. Ed Cox, Executive Vice President, Strategic Alliances & Global Head of Digital Medicine at EVERSANA, interviewed Anish Shindore, Head of Digital […]

Digital Innovation in Asia Pacific
Digital medicine and digital therapeutics are transforming the healthcare industry by changing the care delivery format, and they have the potential to improve patient adherence and outcomes. In our recent Digital Symposium, Ed Cox, Executive Vice President, Strategic Alliances & Global Head of Digital Medicine at EVERSANA, interviewed David Keene, CEO at IntroSpect Digital Therapeutics […]

Outlook: Digital & Pharma in Asia Pacific
Asia Pacific is one of the fastest-growing regions for digital innovation in the pharmaceutical and biotechnology industries. Its promising outlook is mainly driven by necessity, commercial flexibility and a broad talent pool. In a candid conversation broadcasted during the EVERSANA’s Digital Symposium, Ed Cox, Executive Vice President, Strategic Alliances & Global Head of Digital Medicine, […]

Leading and Decision-Making in Uncertain Times
By definition, leaders must have followers, who are defined as people who must be shown the way. While this is within most leaders’ grasps when the road ahead is straight and wide and the destination is visible, more is demanded of them when the road splits, narrows and dusk falls. The current crisis presents special […]

What kind of clinical data is required for FDA clearance of a Digital Medicine?
What kind of clinical data is required for FDA clearance of a Digital Medicine? It depends. Digital therapeutics and digital medicines that are subject to FDA regulation are considered medical devices. The amount of clinical data required by FDA generally depends on risk. Many moderate risk (class II) medical devices seeking 510(k) clearance are required […]

What specifically triggers FDA regulation of a digital medicine?
I often meet with Digital Health or Pharmaceutical clients to discuss their commercialization journey, and they ask me the same question. What specifically triggers FDA regulation of a digital medicine? Some of these products require FDA clearance and some do not. Regulatory oversight of digital medicines depends primarily on intended use, which is generally captured […]

Validate and Inform your Account Access Strategies and Tactics
With $250 million in product revenue, our client is the U.S. subsidiary of a European niche biopharmaceutical company looking to build its presence in the U.S. marketplace in anticipation of future product launches. See how the Health Strategies Insights by EVERSANA Team helped the client put an account strategy in place for each key payer […]

Realizing Value from Digital in the Healthcare Environment [Webinar]
[WEBINAR – 20 MINUTES] The advancements of Digital Therapies are promising but commercial success is not always guaranteed. This presentation explores the pitfalls and challenges of selling products into the uniquely complicated and regulated healthcare market. It will lay the groundwork for digital companies to have successful commercial strategies and be able to: Answer questions […]

The New Roaring 20s: How Outsourced Commercialization is the Revolution We Need
The “Great Gatsby Era” was an exciting time of new prosperity, infinite creativity and dramatic social change. Long-standing social norms and traditions gave way to the “mass culture” and consumerism that modernized American society. “What will they think of next?” was a popular expression that defined the era. On the 100-year anniversary of this revolutionary […]

Improving the Impact of an Oncology Patient Access Program
Our client, a specialty pharmaceutical company focused on the research and development of new therapies in two high-cost complex drug categories, sought to benchmark its patient assistance program against those of its competitors in order to improve relationships within target oncologists’ offices. The Challenge The client needed to assess the image of its patient assistance […]

Strategies to Ensure Market Success of a Rare Disease Product Launch
Small patient populations, complex administration, high costs of therapies, and government policy interventions are just a few obstacles in rare disease. EVERSANA’s Managing Director, Bill O’Bryon, and Senior Director, Diann Johnson, outline key insights to consider in your launch strategy: Market dynamics impacting rare disease therapies The entire rare community: patients, caregivers, providers & advocates […]

After FDA Approval, Can Gene Therapies Achieve Marketing Success?
Without a doubt, gene therapy is transforming healthcare by revolutionizing patient care from conventional treatment models to curative therapy models. There are more than 7,000 distinct types of rare and genetic diseases and 400+ million individuals suffering from a rare disease. With a market that is fragmented, there is a need for an innovative, end-to-end […]

Global Biosimilar Competitor Assessment – Who Wins in Biosimilar Access and Why?
The introduction of biosimilar products has created interesting changes in payer behavior across the global markets. For both innovator and biosimilar companies, it has been an effort to understand the demands of payers and succeed in what looked like a ‘winner-takes-all’ opportunity. As the first round of biosimilar introductions has passed, there are some interesting […]

How do we pay for a cure?
Setting drug prices is a high-stakes endeavor, and that is especially true for the latest round of gene and curative products coming to market in Europe. Drugmakers need to consider several factors that can help them set their product prices appropriately, including the risk of recurrence/relapse, differences in efficacy among cures, and comparators, including full […]

Innovative Therapies Call for an Integrated Drug Safety and Compliance Model
In this article, Herbert Lee, PharmD, Senior Vice President, Medical Communications and Pharmacovigilance, makes the case for an integrated approach to ensuring the safe and effective use of medications by patients. From emerging therapies to innovative technologies, the healthcare industry is changing – demanding that we do business differently. Working in one of the most […]

Navigating Channel Distribution in a New (and more complex) Biopharma World
Every twist and turn in the bench-to-bedside commercialization journey introduces increasing regulatory complexity, product-specific special handling requirements, and evolving industry guidelines. With more than 300 client case studies to draw from, we’ve found three opportunities (or pitfalls) that manufacturers must address in their channel distribution strategy: How quickly can you get your product into your […]

Are We Transforming In The Right Way?
Why Product Launches Can’t Be Distracted By Empty Promises. We can all agree that innovations in therapeutic development have advanced beyond traditional product launch strategies and service models. In every step of the product lifecycle, we see pockets of transformation. The problem is exactly that – “pockets” of transformation. Your product launch strategy has to […]

Gene Therapy for Rare Disorders
With only three approved products in the marketplace, how do we build a regenerative medicine ecosystem that delivers more value to patient’s faster?
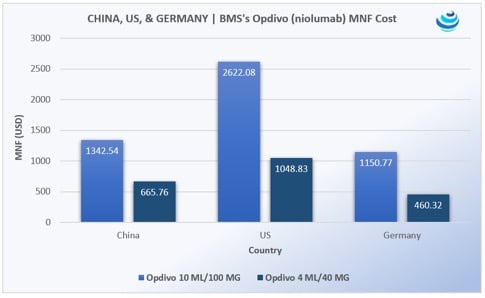
‘Tis the season for pharma in China, as the government expedites uptake of new urgently needed drugs
PRICENTRIC BRIEF: Universal health insurance coverage for 1.3 billion people means China must increase uptake of “clinically urgently needed new drugs” already approved in the US, EU, and Japan Mostly oncology products are being imported, along with HIV/AIDS medicines Pharma is agreeing to steep discounts to be reimbursed on China’s national drug lists THE DETAILS […]
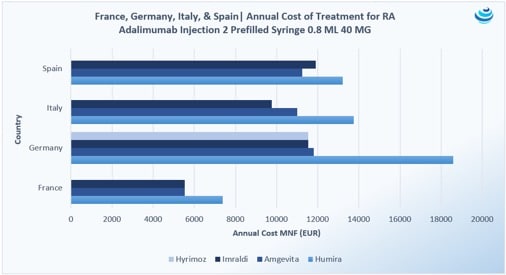
“Who wants the biggest slice of the biosimilar pie?”: The Humira biosimilar wave in Europe
PRICENTRIC BRIEF: Biosimilar competition in Europe has brought about discounts to AbbVie’s blockbuster immunosuppressant drug Humira upwards of 80% during tendering Overall, biosimilar uptake has increased in Europe because biologic “copycats” are cheaper, but full faith in these products is still required from physicians and patients In the US, Coherus struck preemptively with a 33% […]

Biosimilar Pricing in Europe
This report titled Biosimilar Pricing in Europe is published by Pricentric, by EVERSANA. It examines the pricing and pricing trends of biosimilar drugs in the US and EU5. It particularly looks at the pricing of Infliximab and its effect in the markets.

The Brink of the Biosimilar Revolution
Although biosimilars have not had the immediate uptake in the United States that they have had in Europe, health plans expect these products will become a staple of therapy for some diseases by 2020—if the price is right. Since the first biosimilar was approved in the United States in 2015, brand marketers have been bracing […]

