
NAVLIN Price & Access Data
NAVLIN Price & Access Data is a powerful competitor intelligence tool that provides near real-time updates to drug price (list / net), reimbursement, tender and cost of treatment information at the indication level.
Our team of experts track real time pricing changes across 87+ markets and 200+ therapeutic areas and develop county profiles to inform global pricing and market access strategies for our pharma clients.
Articles by NAVLIN Price & Access Data
Global Regulations Push Accelerated Approvals, Access and Treatment Distribution: June 2021
Improving patient access and treatment affordability is quickly becoming a global priority reflected in current regulatory actions. This month, the U.S. FDA approved Aduhelm (aducanumab) for Alzheimer’s through an Accelerated Approval pathway, which will open treatment opportunities for patients. Meanwhile, the world continues to push on in the fight against COVID: The EU is proposing a multilateral trade action plan to promote broad production of and fair access to COVID-19 medications, and the U.S. bought 200 million additional doses of Moderna’s COVID-19 vaccine. Patients around the world are also receiving novel treatments for the first time, such as a five-month-old baby receiving Zolgensma in the UK, as countries work to make generic and novel drugs more accessible. Learn more about global pricing and government regulations in the latest Pricentric ONE by EVERSANA™ INSIGHTS Newsletter. Here are a few of this month’s top stories: […]

INFOGRAPHIC: What You Need To Know About The Tender Landscape In Europe
In today’s market, manufacturers need real-time pricing insights to remain competitive and develop strategies supported by the right data. PriceRight® by EVERSANA is helping manufacturers manage enterprise and government pricing changes for markets around the world, which is critical as manufacturers launch globally. More specifically, manufacturers launching in Europe must be up to date on regulatory changes, including those related to Brexit and the Pharmaceutical Strategy for Europe. To help deliver key insights to our clients, the PriceRight team surveyed 21 European countries about their price regulation and tendering approach for generic medicines. Talk to one of our pricing experts today. Download some insights from this survey now!

Global Pharma Heads Into Uncharted Territories in Pricing and Regulation: May 2021
Policymakers around the world continue to face unprecedented decisions about pharmaceuticals and the healthcare industry. With rapid industry innovation comes new challenges, unanswered questions and intense debates on pricing and regulation. This month, the industry saw progression in rare disease education for policymakers, a spotlight on drug price transparency bills and pricing penalties, advancements in digital therapeutics, development in global HTA use and continued efforts to fight the COVID-19 pandemic. Here are a few of this month’s top stories: Pricentric INSIGHT: BeNeLuxA’s Push for Innovative Drug Price Reduction Sees Zolgensma, Vertex’s CF Portfolio in Firing Line At the start of May, the Netherlands’ Zorginstituut (ZIN) advised the country’s Minister for Medical Care to only reimburse Novartis’ Zolgensma (onasemnogene abeparvovec) […]

Beyond the New Normal: Reshaping Life Sciences Pricing Strategies in a Complex Global Scenario
This Life Sciences Dynamic Talks session takes a close look at the future of life sciences pricing strategies in the “new normal.” From non-traditional blocs to Brexit and the rise of net price transparency, a number of global trends exacerbated by COVID-19 are increasingly affecting pricing for pharma. In this session, EVERSANA’s Alan Crowther identifies recent and upcoming changes to the global pharmaceutical market around launch sequencing and net price transparency and discusses how these changes will affect product launches. He also highlights how companies can prepare for future launches in this new and shifting market.

Pharma’s Push and Pull: Products Approved Amid Government Pushes for Regulation and Pricing Change: April 2021
Global healthcare and life sciences innovation continues to surge at an unprecedented rate, and government entities worldwide are trying to keep up. Price transparency and treatment affordability remain at the top of the list for legislative concern. For instance, United States Senator Jeff Merkley (D-OR) and Representative Peter Welch (D-VT) have introduced the End Price Gouging for Medications Act, which would ensure Americans pay no more for prescription drugs than the median price found among 11 reference countries. United States Representatives David B. McKinley (R-WV) and Annie Kuster (D-NH) have also reintroduced the Ensuring Access to Lower-Cost Medicines for Seniors Act, which aims to leverage generic drugs to help Medicare beneficiaries with affordability. Additionally, health authorities from Argentina, Brazil, Bolivia, Paraguay and […]

The Secret’s Out: The Impact of Net Price Transparency
Pricing system pressures are increasing for the top four markets in Europe. Price transparency, changes to public health plans and curative therapies are set to disrupt traditional drug pricing. To navigate this ever-shifting landscape, companies must carefully consider product adoption and launch sequencing, and understand the potential impact that net price transparency might have. In this webinar, EVERSANA’s Alan Crowther, General Manager, Global Pricing and Market Access Solutions, and Ed Corbett, Senior Principal, EVERSANA™ CONSULTING, EMEA, discuss: Situation: What’s the current global landscape of net price transparency? Implications: How are the changes going to impact current in-market products and future product launches? Actions: What actions do firms need to take […]

Global Shifts in Legislative Regulation Are Forcing Pharma Back to the Drawing Board : February 2021
In only a short time this year, the pharmaceutical industry is witnessing new regulations and legislative changes going into effect globally – and the growing pains are setting in. Between the new U.S. administration and E.U. Pharmaceutical Strategy, governmental entities are starting to set new pricing standards and expectations. The rise of net price transparency, U.S. reference pricing and the prioritization of launch in small markets have all contributed to international regulation trigger pulls – not to mention that China’s pharma industry and non-traditional blocs, such as ACSS and ORBIS, are competing for space in the global market, driving pricing as well. Manufacturers are already beginning to see the rewards of adapting to new regulations quickly, as well as the firestorms that result from stale strategies and tactics that fail to prioritize affordability and access. Read the latest Pricentric ONE INSIGHTS newsletter for global pricing trends […]

A Time for Transition in Pharma: January 2021
So far, 2021 is proving to be a time for international transition in pharma. In the latest Pricentric INSIGHTS newsletter, we’re taking a closer look at new legislation and regulatory actions happening around the globe to help you prepare for what’s coming next this year. Here are the top stories from January: Trump’s MFN Rule Roll-Out Stalled by California, Maryland Courts: District court judges in Maryland and Northern California have ruled to stall the implementation of President Donald Trump’s “Most Favored Nations (MFN)” rule that was supposed to take effect on January 1, 2021. BMS, Roche, AZ Lose Out as China Favors Domestic Companies in NRDL PD-1 Battle: In the […]

A Rundown of 2020 Trends in Pharma
As we begin 2021 and vaccines roll out across the globe, it seems that the world is hopeful and eager to move forward from 2020. The COVID-19 pandemic impacted almost every aspect of our lives, wreaking havoc on numerous industries and forcing global companies to re-evaluate their future. But one industry that was consistently in the spotlight was the pharmaceutical industry. The world watched as pharma players collaborated to end the pandemic as quickly as possible. As a result, 2020 was also a year of unprecedented innovation and acceleration. From the COVID-19 pandemic and the ensuing public health crisis to major political events, such as Brexit and the 2020 U.S. election, we share with you our latest Insights newsletter. Download our full report to read more about our major learnings from 2020 and what […]

How Does the Biden Administration Want to ‘Take on Pharma?’
Throughout his campaign, Biden has promised to, once in office, expand access to health care through the “Biden Plan,” an evolution of former President Barack Obama’s Affordable Care Act, dubbed “Obamacare,” while “taking on big pharma” and reining in “runaway” drug prices. As with Trump, Biden seeks to leverage the negotiating power of Medicare when determining drug prices, and as detailed on his campaign website, Biden has expressed support for previous legislative efforts to benchmark drug prices against those found in economically similar countries around the world. Newly elected Vice President Kamala Harris is also in favor of adopting a reference price scheme in the U.S., as while running for […]

Do Germany’s New Hemophilia Regulations Have Wider-Reaching Implications?
In July 2019, the Drug Safety and Supply Law (GSAV) was passed by Germany’s Higher Chamber of Parliament, bringing about substantial changes to Germany’s health policy. Besides introducing new strategies for biosimilar uptake and cell and gene therapy monitoring, GSAV provides for more stringent control of the distribution of hemophilia products. Download the FULL Pricentric Insights HERE. Learn more about Pricentric ONE and our Global Pricing Solutions! Contact us with your questions and global pricing needs, and an expert will follow up shortly.

Italy Tackles HTA, Pricing and Reimbursement Reforms
In July, new legislation set to overhaul the way pharmaceuticals are priced in Italy was finally implemented, following an announcement that dates all the way back to August 2019. The provision, which was signed by then Ministers Giulia Grillo (Health) and Giovanni Tria (Economy), has rolled out new criteria and methods by which the Italian Medicines Agency (AIFA) determines, via negotiation, the prices of drugs reimbursed by the national health service (SSN). Italy finally rolled out the new policy this year, replacing the “Delibera CIPE” that had governed the Italian pricing and reimbursement of medicines since 2001. Download the FULL Pricentric Insights HERE. Learn more about Pricentric ONE and our […]

At the COVID-19 Finish Line, How Do We Price The Winning Vaccine?
As of August 21, 2020, there were more than 165 potential vaccine candidates in development for the novel coronavirus. While some are still in preclinical stages, a handful are quickly making their way toward regulatory approval, meaning that pricing is starting to become an urgent reality. The first safety trials in humans started as early as March, due to various expedited clinical development processes in place around the globe that promised to bring a vaccine to fruition as quickly as 12 to 18 months from the outbreak of the pandemic—an unprecedented time frame that was quick to spark a reaction from critics. Download the FULL Pricentric Insights HERE. Learn more […]

Is BeNeLuxA Equipped for a Zolgensma Assessment?
The BeNeLuxA initiative is set to take on Zolgensma (onasemnogene abeparvovec), an innovative gene therapy for children under two years old with spinal muscular atrophy (SMA), which was granted conditional approval for use in Europe in May. Belgium, Ireland, and the Netherlands will undertake a joint health technology assessment (HTA) of Zolgensma as part of the application from AveXis, a Novartis company, with Austria serving as expert reviewer. The participating national HTA bodies are committed to aligning their timelines, methodology, and content of HTA processes, and after assessment, the countries will determine if they will enter joint price negotiations. However, whether Zolgensma will be reimbursed is up to the discretion […]
Pricentric ONE Newsletter | June 2020
Good news, this month’s issue of the Pricentric Newsletter is now available for download. Download the Newsletter!

What Canada’s IRP Changes Could Mean for the Industry
Last August, Canada’s Patented Medicine Prices Review Board (PMPRB) announced a set of amendments for its regulatory measures, in a bid to strengthen and modernize the country’s pricing framework for patented drugs. Following the initial proposal of the alterations, the PMPRB released its new draft guidelines in November 2019 and launched a 60-day consultation period with stakeholders and interested members of the public. The amendments, which empower the PMPRB to more strictly control drug prices and reorganize Canada’s reference basket countries to include markets where drug prices are lower, received serious industry backlash, with certain pharmaceutical companies warning that Canada could experience severe launch delays, among other repercussions, if it […]
Pricentric ONE Newsletter | May 2020
Good news, this month’s issue of the Pricentric Newsletter is now available for download. Download the Newsletter!

Pricentric ONE Newsletter | April 2020
Good news, this month’s issue of the Pricentric Newsletter is now available for download. Download the Newsletter!

Is ICER Increasing Influence in the U.S. Over Time?
Recent high-profile product launches have reignited the debate on healthcare costs in the US. The Institute for Clinical and Economic Review (ICER) is an independent research organization that determines value-based price benchmarks. In a recent study, Dr. Stino, Product Manager, examined recent high-profile launches to understand the interaction between ICER value-based price benchmarks and actual launch prices. Read more, download the report: Learn more about Pricentric One and our Global Pricing Solutions! Contact us with your questions and global pricing needs, and an expert will follow up shortly.

How do we pay for a cure?
Setting drug prices is a high-stakes endeavor, and that is especially true for the latest round of gene and curative products coming to market in Europe. Drugmakers need to consider several factors that can help them set their product prices appropriately, including the risk of recurrence/relapse, differences in efficacy among cures, and comparators, including full lifecycle costs of a disease and the burden of health economic costs on budgets. They also need to design innovative payment models that will be attractive to health systems facing increased budgetary pressures as more gene and curative products enter the market. This includes outcomes- and performance-based schemes, which have gained momentum across Europe. However, […]
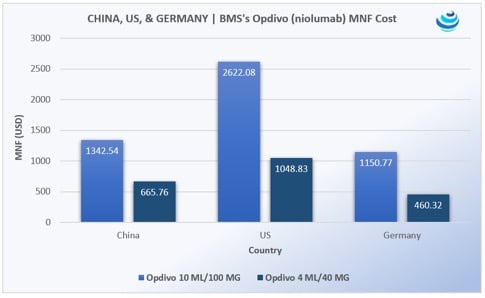
‘Tis the season for pharma in China, as the government expedites uptake of new urgently needed drugs
PRICENTRIC BRIEF: Universal health insurance coverage for 1.3 billion people means China must increase uptake of “clinically urgently needed new drugs” already approved in the US, EU, and Japan Mostly oncology products are being imported, along with HIV/AIDS medicines Pharma is agreeing to steep discounts to be reimbursed on China’s national drug lists THE DETAILS In 2016, China announced that it would be merging the New Rural Cooperative Medical Scheme (NRCMS) and Urban Employee Basic Medical Insurance (UEBMI) to form an integrated system of basic medical insurance for both rural and urban residents, as part of the 2009 Health Care Reform Plan, which seeks universal coverage for 1.3 billion people […]
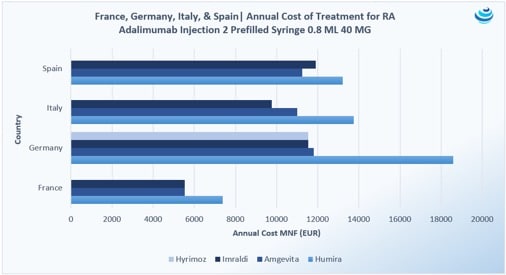
“Who wants the biggest slice of the biosimilar pie?”: The Humira biosimilar wave in Europe
PRICENTRIC BRIEF: Biosimilar competition in Europe has brought about discounts to AbbVie’s blockbuster immunosuppressant drug Humira upwards of 80% during tendering Overall, biosimilar uptake has increased in Europe because biologic “copycats” are cheaper, but full faith in these products is still required from physicians and patients In the US, Coherus struck preemptively with a 33% discount to its Neulasta biosimilar to ensure market uptake THE DETAILS Health authorities the world over seek serious discounts when biosimilar competition enters the market, in many cases mandating discounts and price ceilings to both the originator drug and biosimilars or engaging in tendering practices that allow originator drugs and biosimilars to bid at a […]

Biosimilar Pricing in Europe
This report titled Biosimilar Pricing in Europe is published by Pricentric, by EVERSANA. It examines the pricing and pricing trends of biosimilar drugs in the US and EU5. It particularly looks at the pricing of Infliximab and its effect in the markets.

