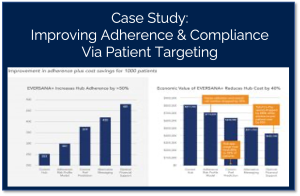
Pharma Companies have over-invested in data and under-invested in actionable insights.
Pharma leaders once thought that more data would lead to greater strategic insights. However, when you ask key decision makers, they’ll tell you they now feel overwhelmed by the amount of data at their fingertips and by the time it takes to glean actionable insights.
EVERSANA’s suite of Data & Analytics solutions, powered by ACTICS by EVERSANA, shortcut the data management and analysis processes for our clients – quickly delivering the right insights to inform and guide strategic decisions that amplify impact across the commercial eco-system.
It all begins with discovery.
EVERSANA will assess and audit what data, infrastructure and tools are needed to answer your specific business questions, providing: